TIN-Bismuth ON STAINLESS STEEL. BRILLIANT
Description
Tinning of stainless steel with a tin-bismuth alloy solves three main problems: protecting the surface from direct contact with aluminum and copper (corrosive galvanic couple), improving solderability, facilitating screwability with simultaneous sealing of threaded connections. Tin plating is also done on marine grade stainless steel parts. An underlayer of nickel 1-2.5 microns thick before tinning stainless steel is required, because without it, the coating will peel off. The corrosion rate of pure tin depending on the pH of the medium is shown in the figure:
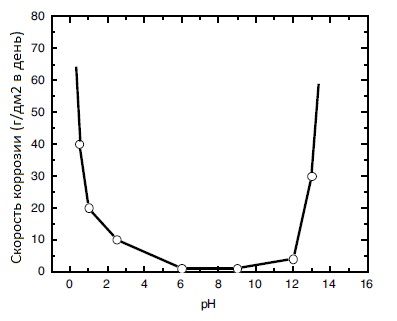
You can order shiny tinning of stainless steel with tin-bismuth alloy in accordance with GOST 9.305-84 by phone and email specified in the section "CONTACTS".
Characteristics
|
Designation (example) |
Sn-Bi |
|
Thickness |
3-100 microns (greater thickness is possible) |
|
Microhardness |
118-198 MPa |
|
Electrical resistivity at 18° C |
11.5⋅10-8 Ohm⋅m |
|
Permissible operating temperature |
200° C |
|
Bismuth content in O-Vi alloy |
0.2-2% |
Advantages of tin plating:
- Tin coating with a nickel underlayer increases the corrosion resistance of stainless steel products in a saline environment.
- The tin coating prevents direct contact between stainless steel and aluminum and copper. As a result of such direct contact, it is possible to form a galvanic couple in which stainless steel will act as the cathode and aluminum as the anode, i.e. active corrosion of aluminum will occur. Contact with copper may accelerate corrosion of the steel itself.
- Resistant to sulfur-containing compounds and recommended for parts in contact with all types of plastics and rubbers. Tin is also stable in saline environments.
- Provides solderability of stainless steel. A shiny tin-bismuth coating retains solderability for a longer time than a matte finish. Alloying with bismuth allows you to maintain solderability for more than a year.
- Has good anti-friction properties.
- Doping with bismuth prevents the formation of conductive needle crystals on the coating surface during storage.
- Doping tin with bismuth in an amount of more than 0.5% also avoids destruction of the coating at temperatures below -30°C. The destruction of tin coatings WITHOUT bismuth occurs due to the transition of compact white tin β-Sn into powdery gray α-Sn (the “tin plague” phenomenon).
Disadvantages of tin plating:
- Low wear resistance when rubbing against harder materials and under abrasive influence.
- Lower antifriction properties compared to tin-lead and lead.
- The presence of bismuth in the composition does not allow the use of coated products if they must come into contact with food environments.
- Relatively unstable in an alkaline environment (gradual mild corrosion).

Read also articles
Mechanism of tin plating (tinning)
What are tin and bismuth? Tin plating mechanism and coating structure
Conductive crystals on tin coating
The nature of tin whiskers and the mechanism of their formation

Do you want to become our client?
Just leave your request by filling out the form on the right and we will contact you as soon as possible. Thank you!

By submitting an application, you agree to processing of your personal data. Your data is protected.






